|
This webpage was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
|
What is Hemochromatosis?
Hemochromatosis is the inability of the body to expel excess iron, leading to iron toxicity. Iron is typically used to make hemoglobin, which is the part of the red blood cell that allows it to carry oxygen. As the body already has a hard time removing excess iron, patients suffering from hemochromatosis can quickly have their iron increase to unsafe levels [1].
Symptoms
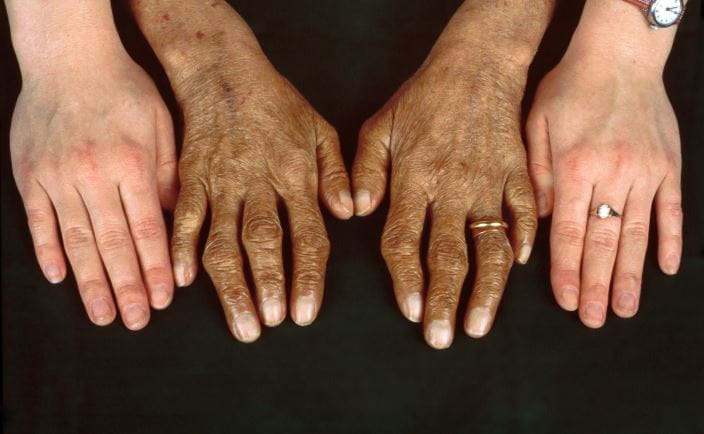
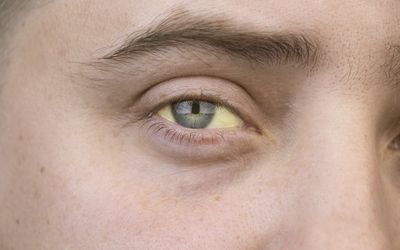
Hemochromatosis can result in bronze or gray skin and hair, fatigue, loss of sex drive, weight loss, and joint and/or abdominal pain. In more extreme cases of iron toxicity, it can cause heart and/or liver damage, liver cancer, arthritis, diabetes, and can be fatal without treatment [2]. Symptoms will begin mildly, and increase over time as iron levels continue to build; they will vary depending on which type of hemochromatosis a person is suffering from [3].
Testing and treatment
Hemochromatosis can sometimes be difficult to initially diagnose, due to the more mild symptoms being fairly common and non specific (such as fatigue). Diagnosis can be achieved via a blood test that measures the amount of iron in the bloodstream, and the amount of iron in the liver [4]. The most common form of treatment is simply donating blood, which is the most direct way to lower iron levels in the body [5]. Other ways to manage iron levels include limiting consumption of red meat and alcohol, and avoiding iron or vitamin C supplements. It is also recommended that raw shellfish is avoided, as individuals with hemochromatosis are more susceptible to a specific bacteria it can sometimes contain.
Types of hemochromatosisEach type of hemochromatosis is caused by a unique gene mutation |
|
Type 1
Caused by a mutation to the HFE gene. Symptoms usually involve scarring of the liver, and begin in mid-adulthood. This is the most common type of hemochromatosis. [2]
|
Type 2
Caused by a mutation to the HAMP gene (type 2A) or the HJV gene (type 2B). Symptoms (which are commonly low levels of sex hormones and heart disease) begin in childhood. [2]
|
Type 3
Caused by a mutation to the TfR2 gene. As symptoms appear around age 30, considered the "intermediate" between types 1 and 2, . Symptoms also typically involve liver scarring. [2]
|
Type 4
Caused by a mutation to the SLC40A1 gene. Least similar to the other three types in terms of what is causing the symptoms. Typically associated with anemia. Age range varies. [2]
|
genetic causes

Each gene that can potentially be mutated to cause hemochromatosis encodes for a specific protein, which has a specific function. If a gene is mutated, then the protein cannot be properly made, meaning it cannot carry out its role correctly, which is how genetic diseases arise. HFE (created by the HFE gene) interacts with another protein called TfR1, which helps regulate iron being brought into cells (fig. 1) [6]. Hepcidin (created by the HAMP gene) helps regulate iron levels by controlling how much is absorbed and how much is released from storage. Hemojuvelin (created by the HJV gene) is shown to help regulate hepcidin, and is theorized to be involved in iron sensing, though its exact role is currently unknown [6]. Neither the biological role nor the function of TfR2 (created by the TfR2 gene) is currently known. Ferroportin (created by the SLC40A1 gene) is responsible for releasing iron stored in cells. A lack of any one of these proteins severely disrupts the highly regulated iron-balance mechanism of the body, ultimately leading to hemochromatosis.
Each of these mutations is autosomal recessive, which means that two copies (one from each parent) of the same mutated gene are required for a child to develop a disease (fig. 2). Unlike most autosomal recessive diseases, however, having one mutated copy of two different genes that have been shown to cause hemochromatosis can sometimes result in symptoms; this has been called an "intermediate" type, as the symptoms and their severity can vary greatly (if they appear at all), depending on what combination mutated genes a person has [6].
Each of these mutations is autosomal recessive, which means that two copies (one from each parent) of the same mutated gene are required for a child to develop a disease (fig. 2). Unlike most autosomal recessive diseases, however, having one mutated copy of two different genes that have been shown to cause hemochromatosis can sometimes result in symptoms; this has been called an "intermediate" type, as the symptoms and their severity can vary greatly (if they appear at all), depending on what combination mutated genes a person has [6].
Figure 2: a diagram demonstrating of autosomal recessive inheritance
References
[1] Billett, Henny. “151.” Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd Edition, LexisNexis UK, 1990.
[2] “Hereditary Hemochromatosis.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 20 May 2022, https://www.cdc.gov/genomics/disease/hemochromatosis.htm.
[3] Fabi, Consuelo, et al. “Hereditary Hemochromatosis Type 3, Analysis of M172K Mutational Sequence in a Single Male Patient.” Biomedres.us, Biomedical Research Network+, LLC, 25 June 2020, https://biomedres.us/fulltexts/BJSTR.MS.ID.004661.php.
[4] “Hemochromatosis.” Mayo Clinic, Mayo Foundation for Medical Education and Research, 6 Jan. 2023, https://www.mayoclinic.org/diseases-conditions/hemochromatosis/diagnosis-treatment/drc-20351448.
[5] “Hemochromatosis.” National Institute of Diabetes and Digestive and Kidney Diseases, U.S. Department of Health and Human Services, https://www.niddk.nih.gov/health-information/liver-disease/hemochromatosis.
[6] Pietrangelo, Antonello. “Hereditary Hemochromatosis.” Biochimica Et Biophysica Acta (BBA) - Molecular Cell Research, Elsevier, 27 May 2006, https://www.sciencedirect.com/science/article/pii/S0167488906001145?via%3Dihub.
[1] Billett, Henny. “151.” Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd Edition, LexisNexis UK, 1990.
[2] “Hereditary Hemochromatosis.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 20 May 2022, https://www.cdc.gov/genomics/disease/hemochromatosis.htm.
[3] Fabi, Consuelo, et al. “Hereditary Hemochromatosis Type 3, Analysis of M172K Mutational Sequence in a Single Male Patient.” Biomedres.us, Biomedical Research Network+, LLC, 25 June 2020, https://biomedres.us/fulltexts/BJSTR.MS.ID.004661.php.
[4] “Hemochromatosis.” Mayo Clinic, Mayo Foundation for Medical Education and Research, 6 Jan. 2023, https://www.mayoclinic.org/diseases-conditions/hemochromatosis/diagnosis-treatment/drc-20351448.
[5] “Hemochromatosis.” National Institute of Diabetes and Digestive and Kidney Diseases, U.S. Department of Health and Human Services, https://www.niddk.nih.gov/health-information/liver-disease/hemochromatosis.
[6] Pietrangelo, Antonello. “Hereditary Hemochromatosis.” Biochimica Et Biophysica Acta (BBA) - Molecular Cell Research, Elsevier, 27 May 2006, https://www.sciencedirect.com/science/article/pii/S0167488906001145?via%3Dihub.